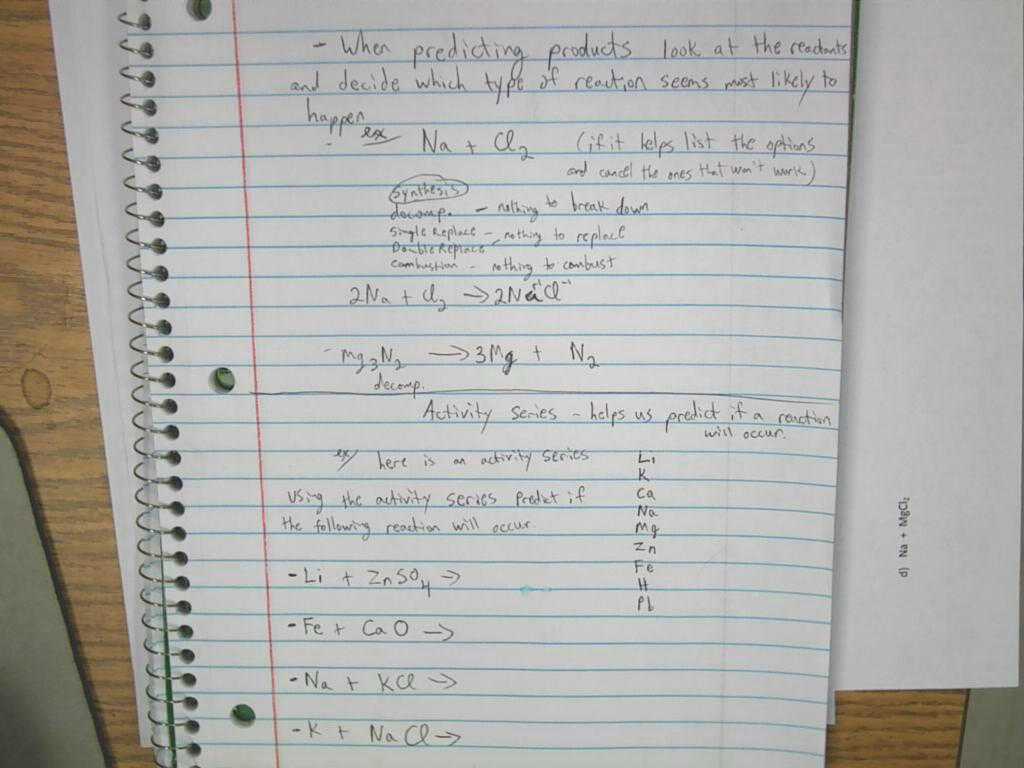Chemistry is a study of matter and its attributes. The word comes from the Greek “charios” which means “craft”. It is therefore an attempt to discover the composition of substances, to identify differences and similarities in properties, shapes, and structures, and so on. The basic idea of the study of matter is that it all has properties of its own, that is it can exist in many different forms, including solid, liquid, or gas.
Understanding of the study of matter is very important for understanding our world. It is based on the fact that all objects, substances, and living things contain some substance that determines their nature. Chemical properties determine the nature of the material and the properties in question can then be classified.

There are two ways in which the study of matter can be understood: firstly, the physical or the way in which substances are found to interact with one another; and secondly, the chemical or the way in which different substances are identified chemically. The chemical properties are determined by the properties of atoms and molecules. Chemical properties also influence the things we call elements. However, in modern terms they are used to describe everything from a particular metal’s color to a flower’s fragrance. It is only in this way that a chemistry student would ever be able to establish that one metal is yellowish and another one is not.
Properties are properties of a substance, whether they are solid liquid or gaseous, and how they interact with other substances. Properties include physical properties, such as shape, volume, or weight, and chemical properties, such as:
The compound – A compound is any group of similar elements joined together in a certain way. For example, iron is a compound of iron, carbon, and oxygen, just as is water. In a compound there is always more than one element present, but they are not necessarily linked together in a definite way.
Molecule – A molecule is a molecule consisting of one atom that is joined to another atom. Molecules can either be single or double, depending on whether they have opposite or similar properties to each other. For example, the carbon atom can be joined to the hydrogen atom to form a single molecule.

Atom – An atom is an atom of matter, a solid, a liquid, or a gas. It is described as being composed of two positively charged parts (electrons) surrounded by a negative part (anions). These are known as the proton and neutron.
There are several examples of elements in chemistry, such as: sodium, potassium, calcium, magnesium, sulfur, potassium, bromine, sulfur, iodine, fluorine, bromine, carbon, iodine, hydrogen, chlorine, sulfur, nitrogen, oxygen, and mercury. Other examples of compounds include silicon, gallium, aluminum, nickel, copper, aluminum, beryllium, boron, fluorine, mercury, lead, and chlorine.