Isotopes are going to have unique atomic mass compared to other atoms which make up the very same element. The isotopes of an element have various masses. Generally, the isotopes of each element have to be given a name that is. Fission wants a neutron to be added to produce the atom unstable. A completely new process would be indicated by seeing a decay, states Kravitz. You should make it clear that decay isn’t utilized by nuclear power stations!
As it produces only water vapor that the moment it 19, 18 Hydrogen gas was suggested as a fuel. It consists of one proton and one electron. For instance, hydrogen contains three isotopes that happen in nature 1H, 2H, 3H. Unique elements’ atoms vary in dimension and mass. Any atom with shells will probably be reactive. Amounts are weighed by atoms. All atoms of the same element that is exact have to possess precisely the same number of protons.
Neutrinos are created while the nucleus of an atom changes in a certain fashion. Ordinarily, there comes a neutrino to a sensor and a lot of the time it isn’t seen by you, Stan Wojcicki states. Neutrinos have a lot of Gratta states, of peculiarities.
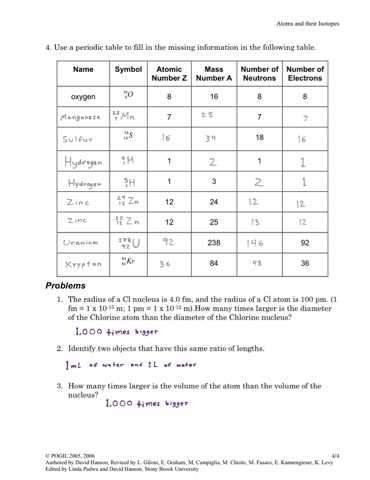
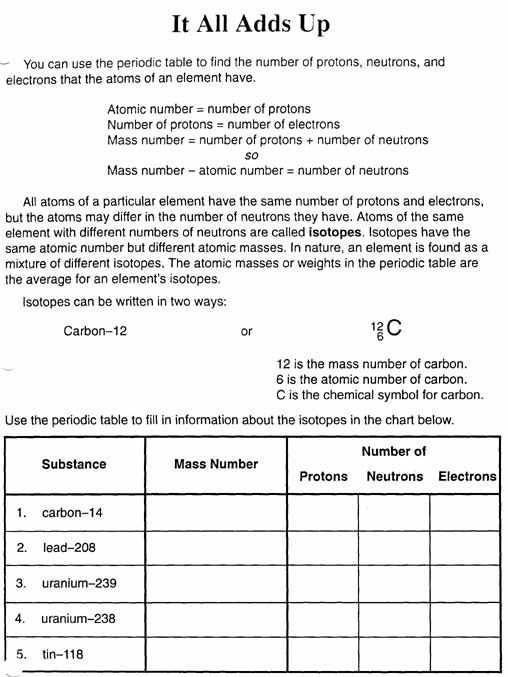
The properties of the elements are dependent on the number of electrons and protons they’ve. The chemical properties of isotopes will be the specific same, despite how some isotopes’ natural features may differ. They are the same although the attributes of some isotopes might differ. The law of conservation of mass is advantageous for many calculations and may be utilized to solve such the quantity of gasoline for unknown masses. Atomic theory was revised to integrate the interconversion of energy and mass as well as the presence of atomic isotopes. Regardless of how scientific models change, versions are the lone way man has to describe how several organic phenomena perform the job.
In a neutral atom, electrons’ array equals the number of protons. The selection of protons is set by the atomic number. In a neutral atom, the array of electrons equals the number of protons. For components, neutrons’ variety isn’t absolutely fixed, unlike protons. To fit the number of neutrons listed on your card into the appropriate element, search for an element on the periodic table in order that in case you add the assortment of neutrons in your card into the protons of this element, you are going to receive near the atomic mass for this element.
There are basically two kinds of sensor today. An element is a substance composed of all the same sort of atom. You may also double-click the template open it and get started customizing it instantly if you find a template that you want to use! The templates in our section will help alleviate a number of the strain which includes such a buy.