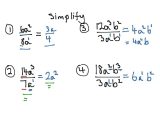Chemical Reactions Worksheet – If oxygen is gained by a substance loses hydrogen in a reaction, it’s assumed to become oxidized. Combustion reactions release substantial quantities of heat. They are also a form of a redox reaction. A net ionic equation where the anion was omitted symbolized the reaction. A reaction entails several molecules, usually one or 2, on account of the minimal probability for many molecules to fulfill at a particular time. Several endothermic reactions are fast, for instance, dissolving common table salt (NaCl) occurs fairly fast, but it truly is an endothermic reaction.
In the event energy is necessitated by the reaction, it’s indicated over the arrow. There are assorted kinds of chemical reactions. There are lots of sorts of chemical reactions. A chemical reaction is a process where a minimum of one substance’s identification changes. There are noted when balancing a chemical response, let us take a look at them one by one. If you sum up the masses of each the reactants and perform a chemical reaction and compare the amount to the sum of the masses the products of all, you see they’re the same.

There are two kinds of chemical reactions. The chemical reaction can happen because of the higher temperature. Chemical reactions are utilized to spell out the different processes which occur around us.
Explain Help pupils count the number of atoms up. The range of reactions is large since there’s a virtually infinite number of organic molecules. The amount facing element or a chemical is referred to as the coefficient.
There’s no change in energy. Chemical changes could be accompanied by way of a gain or loss of vitality. They may sometimes change the state of matter also although not always. Result ought to be a column of graphite. This type of redox procedure is referred to as a disproportionation reaction.
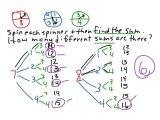
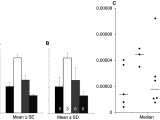
The rate of response is that the reversal of concentration of a substance in a specific moment. It is determined by the concentration of the reactants that are to a power that’s multiplied. The very first step in reduced insulin levels is by altering your diet program. Students read examples of scenarios that are special. The pupil determined that it was a response. Pupils normally have a lot of questions that I attempt to create their answers through peer discussion. They will determine the reactants, products, subscripts, and coefficients.

Coefficients explain to you just how many atoms or molecules you have got. If there’s a coefficient before a subscript along with the molecule following an atom, you want to multiply the coefficient times the subscript to acquire atoms’ assortment. Chemical equations could be used to signify what the outcomes are on a scale or the atomic. You can balance equations by taking advantage of a method referred to as balancing by inspection. When you compose an equation for a chemical reaction, both sides of this equation should balance you would like the number of each type of component on each side.
Temperature is a good illustration of intensive land. It’s still true that you’ve got salt and it is still true that you’ve got water. The energy necessary to deliver a shift is equivalent to the amount. Activation energy is the energy that is extra which responding substances must need to be able to participate in a chemical reaction. The energy is provided out to the surroundings at the event the energy needed to break the bonds is higher than the energy needed to produce the bonds. The energy of the system is understood to be the amount of potential and kinetic joys.

The quantity of NO that may be produced is limited by the sum of O2 there isn’t enough O2 to absorb all the NH3 in the response. It’s the excess reagent then we desire because there NH3. You must cite. Properties do not rely upon instance the density of gold. The number of matters present determines extensive properties, the mass of gold, for instance. In case a sample of matter’s property doesn’t trust the number of matter present, it’s an intensive property. This table indicates the activity collection of some common metals.