It seems as if everyone’s heard of molecular coordination in a simple metal bead or a two-dimensional gold foil, but you might not be familiar with the chemical bonding properties of organic compounds. Ionic and covalent reactions are the building blocks of the organic world – reaction between molecules bound to two or more atoms.
Ionic or covalent reactions are usually one molecule of an ion such as a chloride ion (potassium chloride is good for diabetes patients) or an anion such as an alkali metal or sodium. The covalent type of bond depends on which atoms are bonded in a chemical reaction. When you think of ionic bonding, it is important to remember that the covalent element in a chemical bond, the atom being bonded, needs to be much heavier than the ion to make this covalent bond possible.
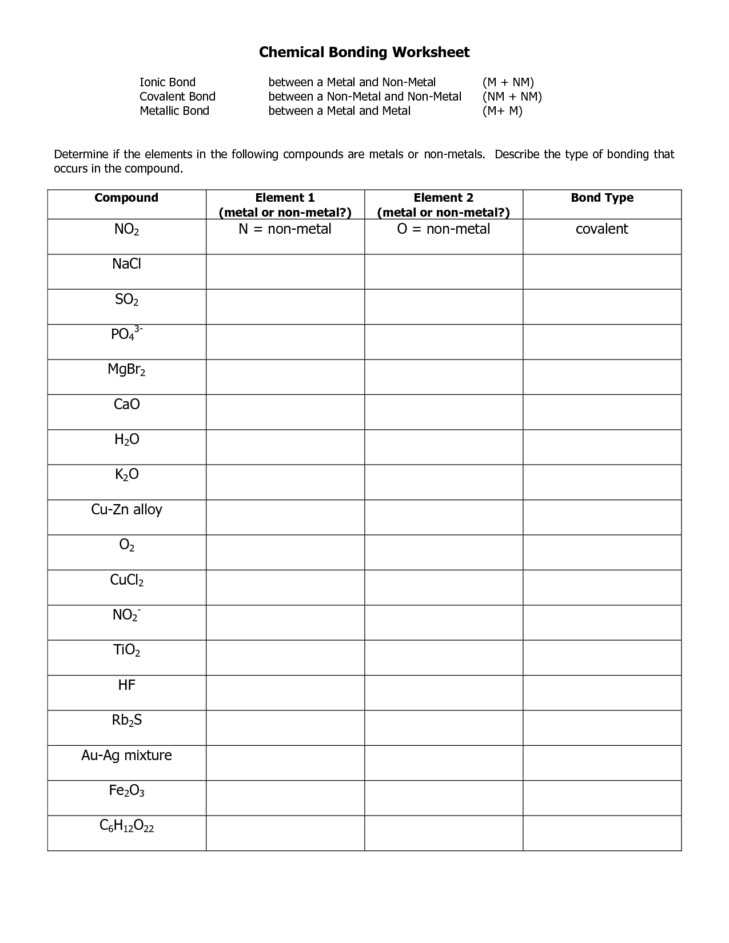
In ionic bonding, the atom of the element is bonded to an atom of the opposite sign. For example, boron atoms bonded to oxygen atoms are covalent. Ionic bonding is often the outcome of the covalent bond, as well. That’s why boron is necessary for the formation of boric acid (boron-borate) in some medical situations.
When it comes to covalent chemical bonds, however, it is really important to get the definitions right. In an ionic chemical bond to the atom of the covalent element (chloride in the sodium example) is bonded to an atom of the opposite sign. Examples of covalent bonds are water molecules bonds to water molecules, and oxygen atoms bound to carbon atoms.
The ionic bonds, when the atom being bonded is potassium ion, which is referred to as potassium and carbon bonds. As for the covalent bonds, we will just call them “chemical bonds” and may remember that they are better for the functionality of the material, but only that.
The processes of the reactions of the covalent with the ionic are called the oxidation and reduction of the chemical. In ionic bonding, the atoms will react so that the substance begins to oxidize. For example, some elements release energy or “substance” when the ion and the ionic form bonds. The energy that is released by the molecule when the two ionic atoms react is a product.

The fact that a molecule will react when it has an ionic bond is not new to us. It is something that all chemical scientists know already, and just as many professionals in other fields that deal with different kinds of chemistry understand it. It is just the difference between the rest of us and the general population in learning the chemical bond in organic compounds.