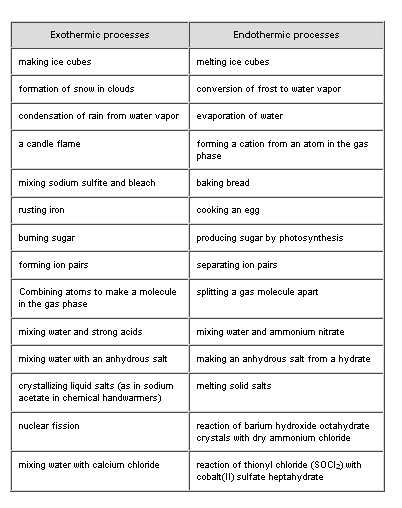
The purpose of this article is to provide you with the answers to your questions about Endothermic and Exothermic Reaction Worksheets. In order to understand these worksheets, you will first need to know what each reaction actually is.
Each reaction has its own name, which is typically based on the type of catalyst that was used in the process. An Exothermic Reaction Worksheet is one that uses an anionic anion in the reaction formula.

The catalyst that is used is commonly referred to as a base. Often times it is another type of catalyst that can be applied to the anionic anion. The catalyst that is used is generally not necessary but may be considered when using other types of formulas.
Thermodynamic Equilibrium is the last step that occurs after all the steps in the equation have been completed. It can be understood with an example as to why the equilibrium is important.
If the hydrogen atom in water reacts with oxygen, then the hydrogen is released because the hydrogen atoms are being replaced by more oxygen atoms. This is considered a Thermodynamic Equilibrium so when a reaction occurs there is a point where the equilibrium is reached.
When the equilibrium is reached, the reaction is finished. Therefore, when you see the endothermic and exothermic reaction worksheet answers listed in the list above, it is here where the equilibrium is reached.
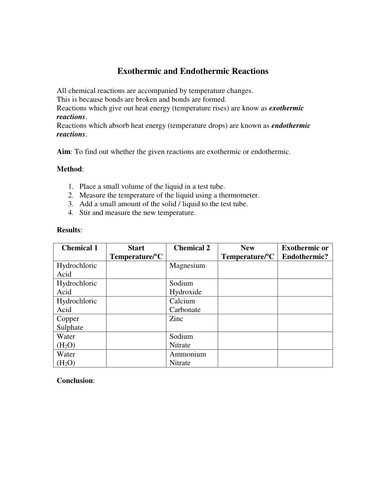
The equilibrium is reached because the ratio of the two ions is equal. The reaction is complete once the ratio has been met.
There are several formulas that are used in these reactions so it is important to know the difference between them. These formulas are in a different order, so it is important to pay attention to the order in which they are listed.