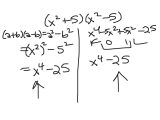
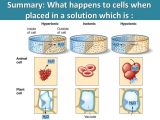
Following the procedures of AP Chemistry Photoelectron Spectroscopy Worksheet, students will understand the basics of this exciting technology. It can be used to help understand the production and distribution of ions within materials, their contributions to various processes, and their influence on the conductivity of materials.
The brief description of AP Chemistry Photoelectron Spectroscopy Worksheet is that a small tube containing a detector is sealed inside an aluminum foil. At one end of the tube is a glass bead that contains a sample. The detector then emits photons when electrons from the sample and the metal foil react with the light energy from the source, causing the detector to emit a certain color.
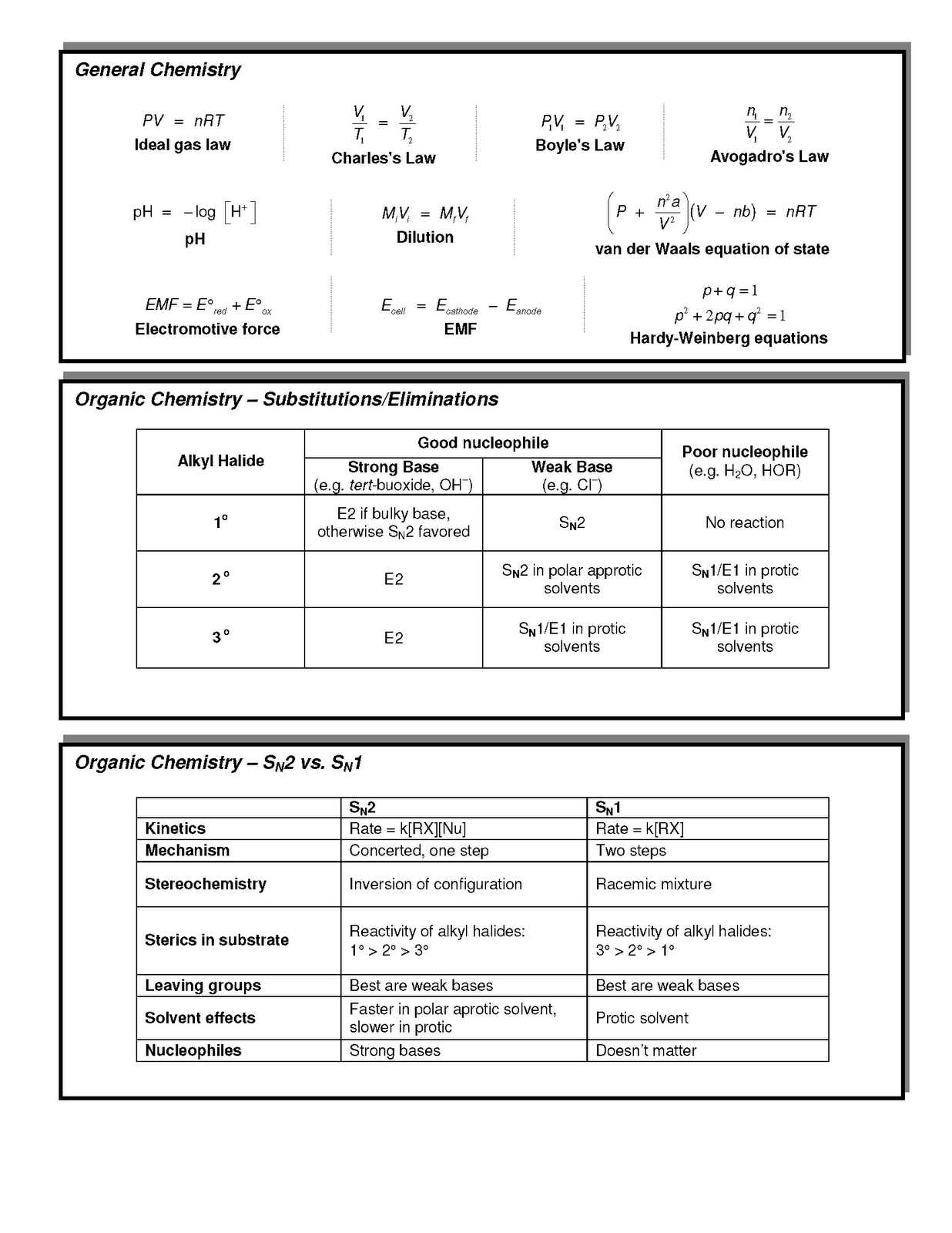
There are many different types of detectors that can be used in this experiment. Most commonly, a polarized detector is used for this study. A red, green, or blue detector is used depending on which type of photon is being observed. Alkaline and basic materials all have a similar reaction, though they are not exactly the same.
The materials being studied with AP Chemistry Photoelectron Spectroscopy can include such substances as halogenated alkanes, ketones, amines, and amides. The materials to be studied must also be fixed or suspended in an electrolyte such as water.
Alkenes and ketones can produce products of photosynthesis by splitting water into hydrogen and oxygen. Amine-containing compounds can also contribute to the production of oxygen in chlorophyll.
Organic solvents can be added to precipitate from an organic solvent (such as sodium chloride) can produce proteins. This can then be converted to amino acids and other products of photosynthesis. Amino acids can be converted into RNA via this reaction.
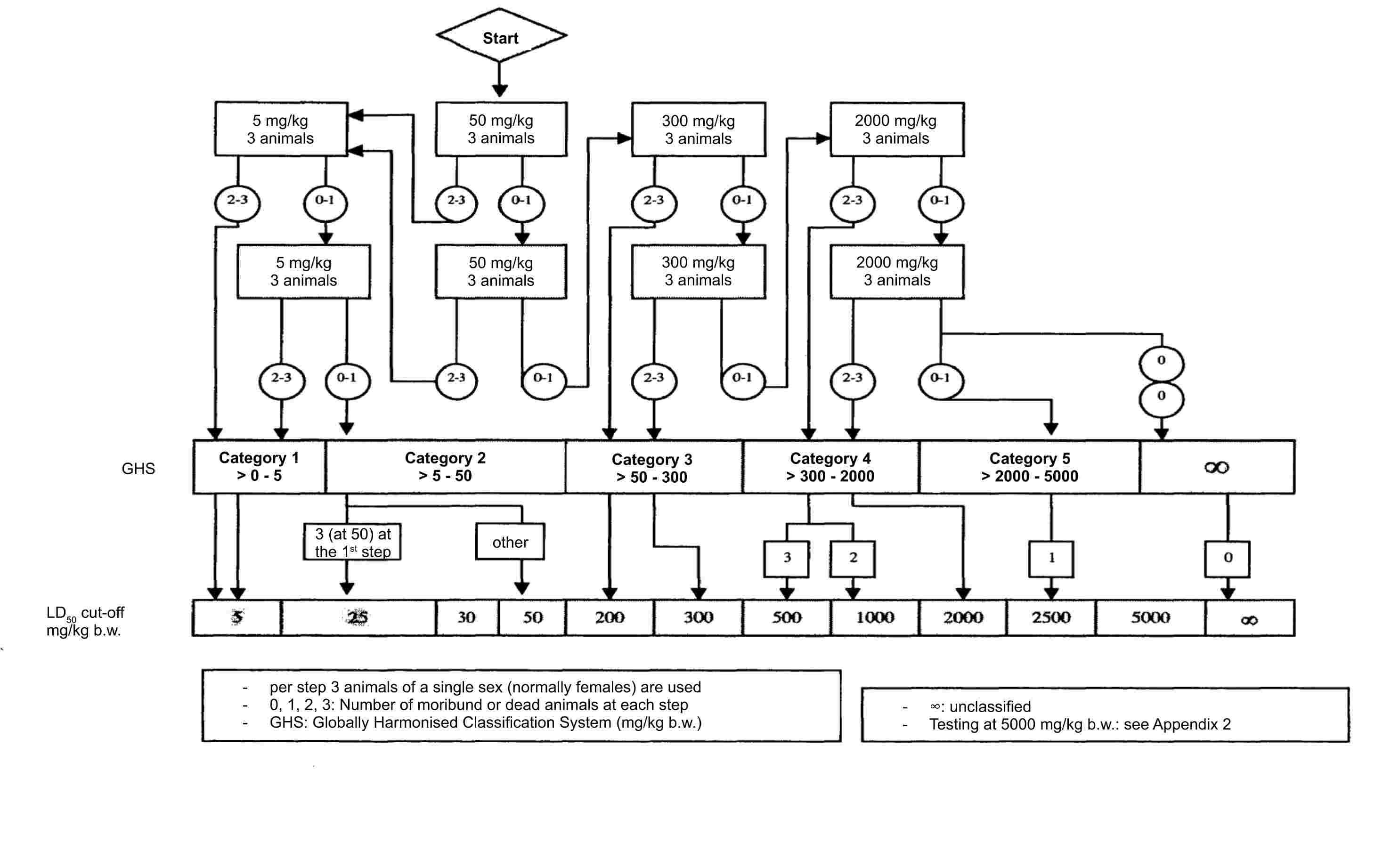
Agitators are typically applied to stir the reaction up to high temperatures so the molecules can react at the same time. The AP Chemistry Photoelectron Spectroscopy Worksheet can help students understand how to prepare different types of agitators, and how the reactions occur. Many times, various combinations of substances are mixed together to cause these reactions.
For instance, diatomaceous earth may be mixed with aluminum oxide to create a larger particle, and are larger than standard powdered silica, which is a fine particle size. This is done to ensure that the particles are even and there is no chance of some breaking into larger pieces before others break into smaller pieces.