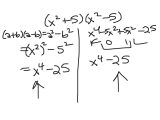
The first four steps of the Chem Worksheet 4 3 to Abundance Of Isotopes gives some background information on the atomic isotope theory. It also introduces the AdS-IS system. This system predicts the properties of a given element based on its number of unstable isotopes and their relative abundance. However, there are some potential problems with this system.
Because isotopes are smaller than their parent isotopes, they will be able to escape from the nucleus quicker. Also, it is possible that different isotopes could end up with different masses.

If these two things happen, then the mass of the parent isotope will increase. That means the decay rate for the unstable isotope increases and eventually it will become too abundant for us to make use of it. We may run out of some elements much sooner than others, depending on how fast these isotopes decay.
However, no matter how many isotopes we make, there are still parts of the planet where they don’t occur at all. There are three main reasons for this: because there isn’t enough uranium or thorium in the ground, because the atmosphere is so high in oxygen that no plant can grow, or because the soil is too dense.
There are two steps to Chemistry Worksheet 4 3 to Abundance Of Isotopes, the first of which is to explain the Isotope Mass Number. The mass number tells us the weight of an isotope when it is similar to its parent isotope. This is very important because it tells us whether there is an abundance of one isotope over another.
It’s actually fairly easy to learn all about isotopes if you read the Materials and Concepts chapter in the book. A better way is to use this Workbook, which has a series of questions on the first page. This makes the first half of the workbook more interesting because you don’t know what the answers are going to be!

The second step in Chemistry Worksheet 4 3 to Abundance Of Isotopes is to examine the Absorption Spectra of oxygen and water isotopes. This is important because it tells us about the nature of chemical bonds. It also explains why atoms don’t have “spikes” in their mass number. The process shows that there must be a limit to the number of atoms in a molecule.
Lastly, in Chemistry Worksheet 4 3 to Abundance Of Isotopes, the process includes analysis of oxygen and water isotopes in some rare cases. There are several ways that isotopes are not produced as frequently as they should be. By studying the work in this book, you can find out which ones are important and which are not.