If you are researching and evaluating the different forms of DNA technology for possible use in manufacturing and diagnostic applications, it is important to understand the information contained in the “Recombinant DNA Worksheet Answers.” This sheet was written by FDA (Food and Drug Administration) and represents the criteria for a biosimilar or product reference. The answers provide a useful guide to evaluating what new DNA products may be introduced into the marketplace and how they might impact a company’s business.
First, the work sheet explains the most critical areas of knowledge when it comes to any DNA technology. It is important to grasp the concept of the DNA molecule (nucleic acid), which describe the gene, and the RNA polymerase, which involve information processing. It explains how these two components work together to create a product’s specific biological functions. These concepts are critical to the identification of genes that are needed for specific applications.
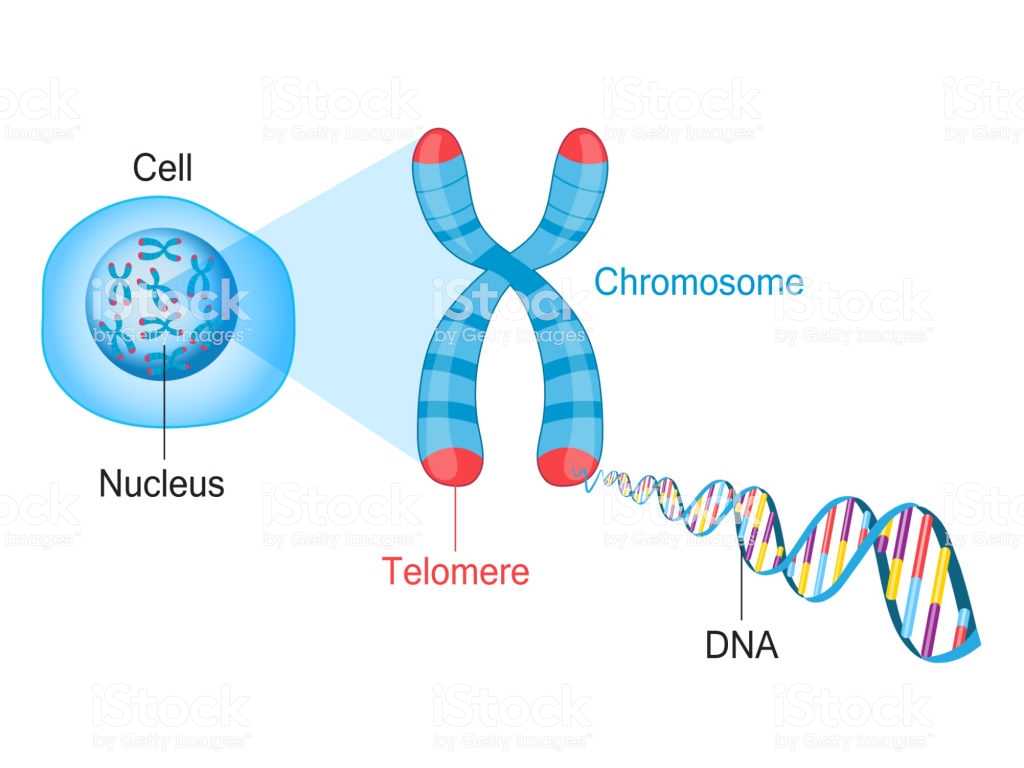
Next, the work sheet answers questions such as, “How do scientists control the use of nucleic acids?” “What are the methods used to reproduce the genetic information?” And “What is the significance of knowing the structural information in a genome?”
While recreating the genetic information has been considered to be a desirable attribute for many DNA applications, one should not have to jump through hoops to use the technology. One should always be able to use the technology to its fullest potential. Some forms of DNA can be used for even more than others.
This is important because many elements in the genome are necessary for practical and functional purposes. For example, functional RNAs are needed for efficient tissue repair and maintenance. These can be used for things such as DNA repair, cell growth, gene expression, and more.
In order to determine if the product being produced by a company is a recombinant DNA, there are a few factors to consider. First, is the product made from a somatic cell? Second, does the product come from a human or animal source? Finally, is the product being produced for therapeutic purposes or for other reasons?

It is important to keep in mind that the DNA used in the creation of a product is considered “modified” DNA. This means that it has some of the original genome, but additional features, structures, and data are added as the company proceeds with their products. The test used to determine whether a product is a modified DNA product is also referred to as the functional application, which refers to whether or not the product is therapeutically significant. For example, if a product is being made to treat cancer, the product is considered functional.
Although the Recombinant DNA worksheet answers are fairly basic, this information is crucial when working with the complex world of DNA technology. Because there are so many issues involved, the body must be thoroughly educated on these issues before allowing any product to be used. This means ensuring that the most current information is available and ensuring that employees have the knowledge to use any new technology correctly. Proper education is imperative to ensure that all employees can do their jobs competently.