If you have recently taken a physics or chemistry course, you may be familiar with the concept of protons and neutrons. In general, all atoms, including water, contain at least one proton and one neutron. In reality, the situation may be more complex than this.
For example, consider the hydrogen atom. Since the hydrogen atom is composed of an electron and an invisible proton, it has an uncharged nucleus. However, its charge is zero because it contains no electrons. Although there are many kinds of atoms, the hydrogen atom is the only one that contains no charge.
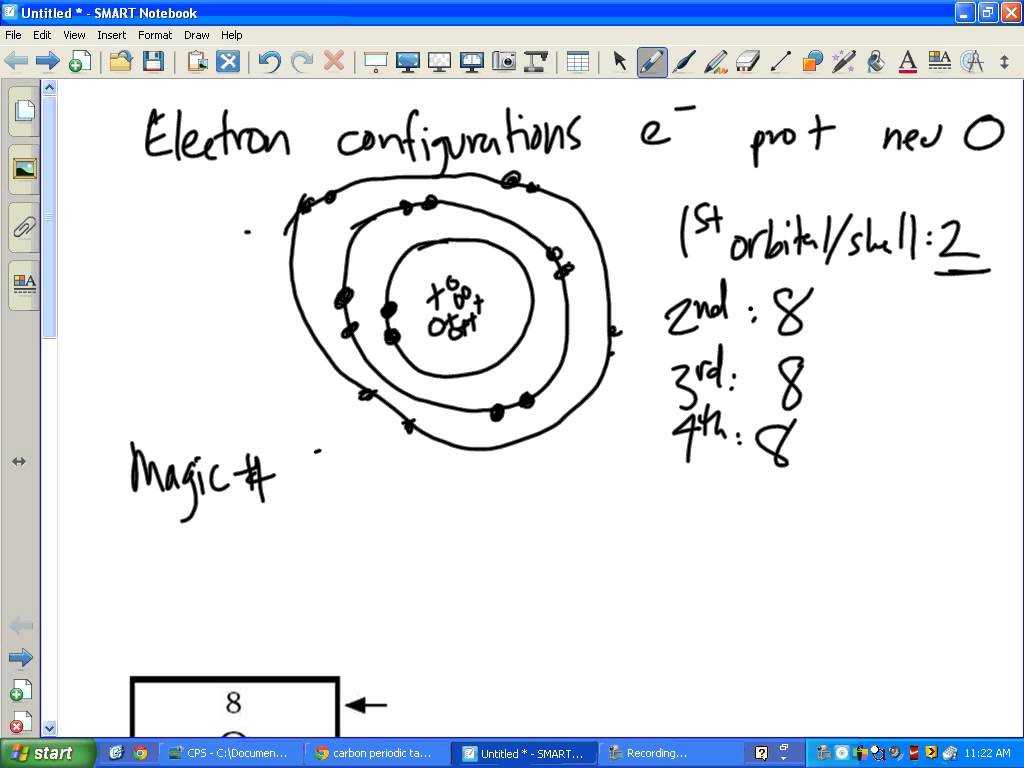
The hydrogen atom is simply an elementary particle of the element hydrogen. Hydrogen is also known as the gas of the element, which is composed of one proton and one electron orbiting each other. It has two isotopes, deuterium and tritium. Trinitrotoluene, or Tilt-L, is a substance which contains hydrogen and nitrogen atoms fused together. It is most commonly used as a chemical marker and dye.
The protons and neutrons of the hydrogen atom are part of an atom’s nucleus. The nucleus is made up of four quarks or gluons which interact through their electromagnetic force to form protons and neutrons. The interaction between these particles is similar to that of a magnetic force, except that the interactions are not created by a particle but rather by the motion of a particle.
The interactions among the protons and neutrons within the hydrogen atom cause them to become electrically charged. Therefore, the proton has an electric charge while the neutron has a magnetic charge. The coupling between the magnetic and electric charges of the proton and neutron causes a drastic change in the momenta of these particles.
The hydrogen atom has two distinct momenta due to the protons and neutrons. One momenta is the positive charge which occurs when protons and neutrons occupy opposite positions. The other momenta is the negative charge which occurs when protons and neutrons occupy the same position. The overall reaction is a phase change of the proton and neutron, which create the effect of a potential.
This is true even though the protons and neutrons do not actually move. They simply change the time it takes for a photon to travel from one position to another. Therefore, each proton and neutron, even though they do not travel, are actually creating a kinetic energy.
This dynamic effect on the atoms is known as the radiation law. This radiation law states that the movement of one proton and neutron produces a relative change in the total mass and velocity of the atom.






