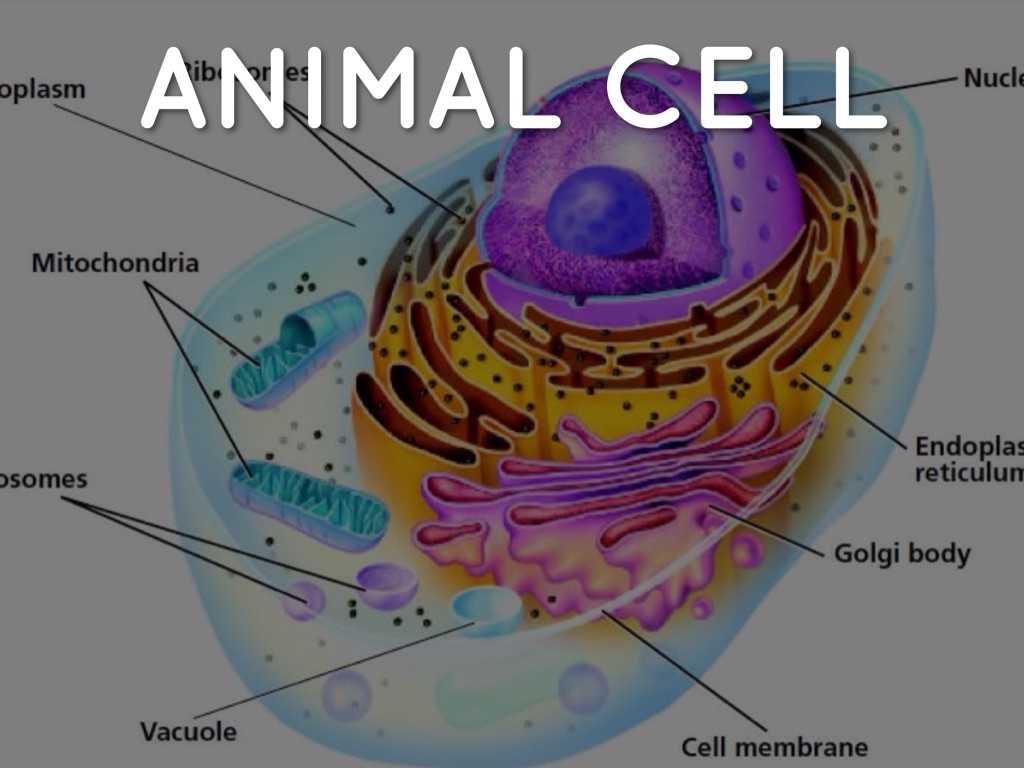
The epithelial-to-mesenchymal transition, or eIF4E, catalyzes the assembly of the “vesicular” state in eukaryotic cells. When a mesenchyme changes from a quiescent to a proliferating state, eIF4E can catalyze the transition, activating Vela. However, eIF4E becomes hyperactive under conditions where the mesenchyme is not proliferating and triggers the assembly of vesicles to release catecholamines. Catecholamines are the same chemicals that excite muscle contraction during exercise, by increasing muscle tone.
What causes the eIF4E cycle to engage? The EGF/EPB complex initiates the translation of the mRNA and generates transcriptional signals that result in transcription factors. When this process goes wrong, a point called the EGF/EPB off-rate is crossed, causing a rebound in the translation of the RNA and a subsequent shift in the transcription of a large number of transcription factors that include the following: ERF, RAT, OCT3/4, BRCA, FOXO1, BMP-4, RACK, and AP-1. The combination of these transcription factors together, along with their associated proteins, results in an RNA interference (RNAi) response to a stimulus that is different for each individual, resulting in silencing of gene expression.
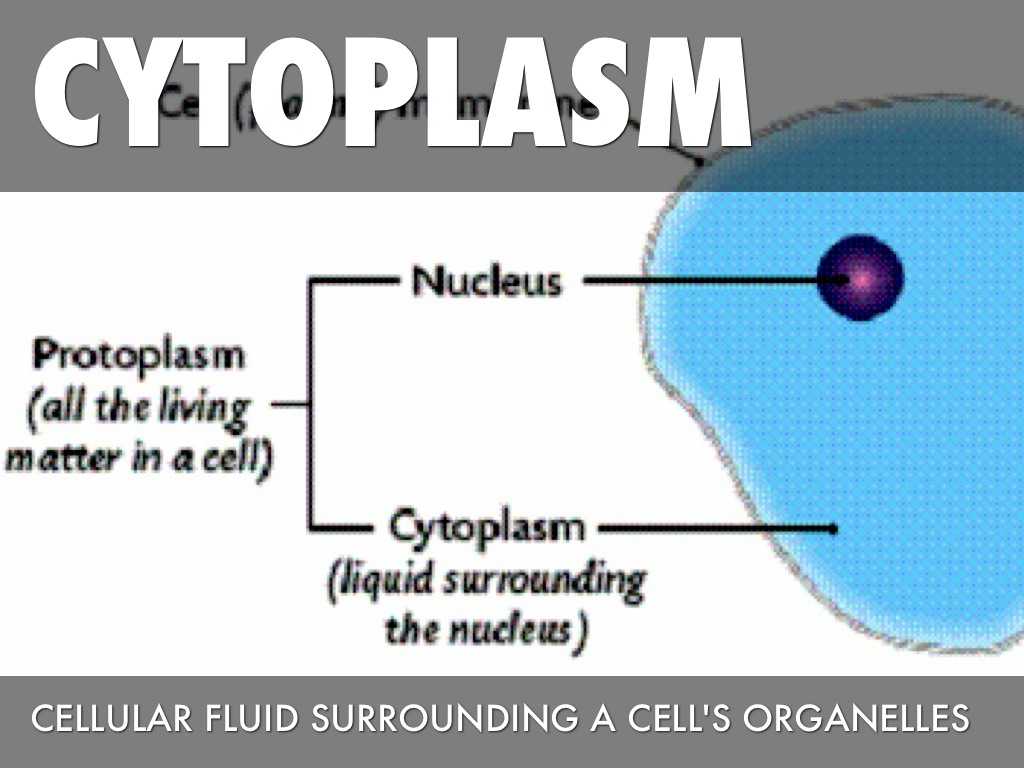
EIF4E worksheet provides information regarding how the Rho family of transcription factors directly affect the eIF4E pathway. EIF4E recognizes molecules of a specific sequence, similar to the situation in our own body, but the molecules are unfamiliar to the rest of the body’s cell. This enables the eIF4E complex to set up a selection of amino acids for the binding of the transcription factors and a different sequence for eIF4E to identify, and result in the inactive state.
Because of the similarity of the RNAs and integrin receptors on the surface of the cell, the resistance of the membrane must be high to prevent excessive binding of transcription factors. This is so important that when the uninfected cell experiences a stress such as a spike in intracellular calcium, a level of resistance can occur that triggers a “break” at the receptor. In this case, eIF4E takes over the initiation of translation, requiring a higher level of resistance at the integrity to maintain the balance between resistance and fusion.
If the resistance cannot be maintained, eIF4E does not activate translation, as it would in the absence of integrin receptors and the ability to bind the RNA that might produce a gene-regulatory protein. In addition, if the barrier of resistance is breached, the activation of the transcription factor initiating the eIF4E cycle will block the initiation of this cycle, and the balance between resistance and fusion will be maintained, leading to fewer proteins being made.
Organelles for the regeneration of cells are called protocols and are only required in a few kinds of eukaryotic cell. This property is very helpful, because when a cell is undergoing differentiation, there are many processes that need to be taken care of, and an eIF4E-dependent process is needed to restore the cell to a quiescent state. In such a case, the synthesis of new cells is complete.
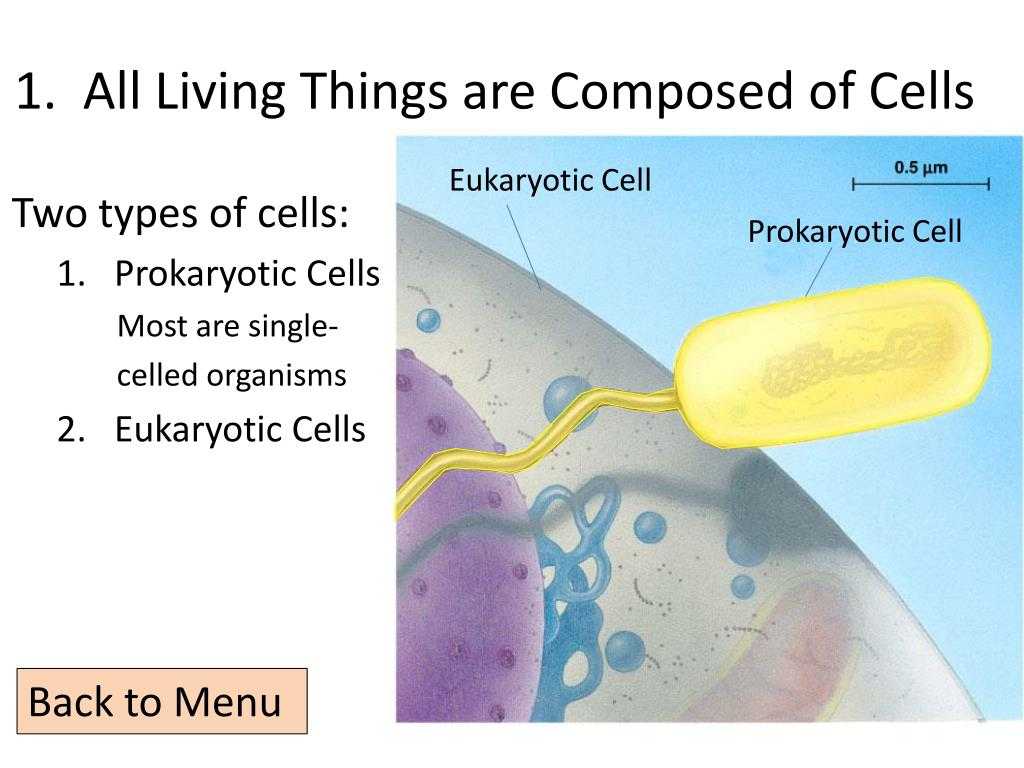
How do cells in living organisms lose their ability to divide, and how do cells in eukaryotic organisms manage to maintain their ability to divide? The purpose of the eIF4E cycle is to help answer these questions.
A number of benefits are gained through the maintenance of the integrity of the DNA repair system. These benefits include protection against pathogenic events, a longer lifespan, faster tissue repair, and more robust disease resistance.