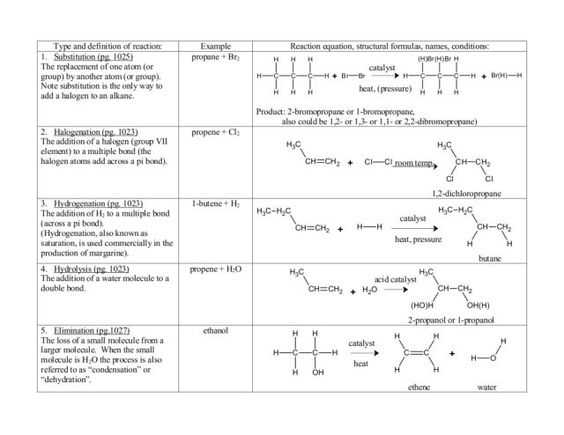
The quantity of energy needed to break a bond is just like the sum of energy released when it forms. Generally, it will be released when a bond forms between two atoms, no matter what type of bond. This energy could be lost as heat or may be used for power. It is just like the energy given out in making the exact amount of covalent bonds. The expression bond energy is usually utilized to spell out the strength of interactions between atoms that produce covalent bonds. It is determined by measuring the heat required to break one mole of molecules into their atoms, and it represents the average energy associated with breaking the individual bonds of a molecule. Bond energies (the quantity of energy that has to be added to be able to break a bond) are an essential element in determining whether or not a reaction will occur.
The precise bond enthalpy of a specific chemical bond is dependent upon the molecular environment where the bond exists. Consequently, their specific bond enthalpies are likely to differ. The bond enthalpy for this situation would differ if six chlorines were instead connected to the carbons.
Bonds between certain specific elements generally have a characteristic energy, known as the bond energy, that’s necessary to break the bond. It makes it a lot less difficult to count up how many of each kind of bond you’ve got to break and make. In cases such as this, the bond enthalpy quoted is a mean price.
Students then utilize words and an energy level diagram to spell out the practice of combustion. With the media, the teacher is necessary to concentrate on giving a maximum comprehension of the designated subject. All you need to do is click the lesson you would like to study and the movie is going to show up on your browser. Worksheets might also be thought to be the use of the problem bank principle to train scholar intelligence. The estimating worksheet was made to direct you. Bond Energy Worksheet is a kind of education aid. Templates might also be helpful as soon as you’re trying to lose or maintain your existing weight.
Should you do an online search, you will discover that other individuals utilize various values for the carbon-carbon single bond. When you arrive in their page, all you need to do is either pick one of several templates they supply or Start Fresh. We’ll see within the next section that there’s another energetic factor, entropy, that we also must consider in reactions.
Enthalpy changes are a remarkably exciting region of practical chemistry that students appreciate. And, the result will produce radicals. The outcome is at the period of evaluation, there’s a whole lot of confusion.

Just use the values you’re given. But this value is lowered substantially in the existence of a solvent. Don’t be worried if you happen across slightly different values. Because it is currently a positive price, it’s the E for those bonds of the product being broken. While breaking this bond the same quantity of energy is demanded.