Should you want, you can go back to the test and make an effort to increase your score. Our quiz and worksheet are composed of multiple-choice questions on molecular geometry to assist you to assess your knowledge. The estimating worksheet was made to direct you get through the estimation practice.
There are a lot of charts excel templates from the world wide web. The molecular geometry chart still applies in the event you own a molecule with over 1 atom. You’re able to create a Lewis electron-dot diagram simply by writing the symbol for the atom you wish to represent.
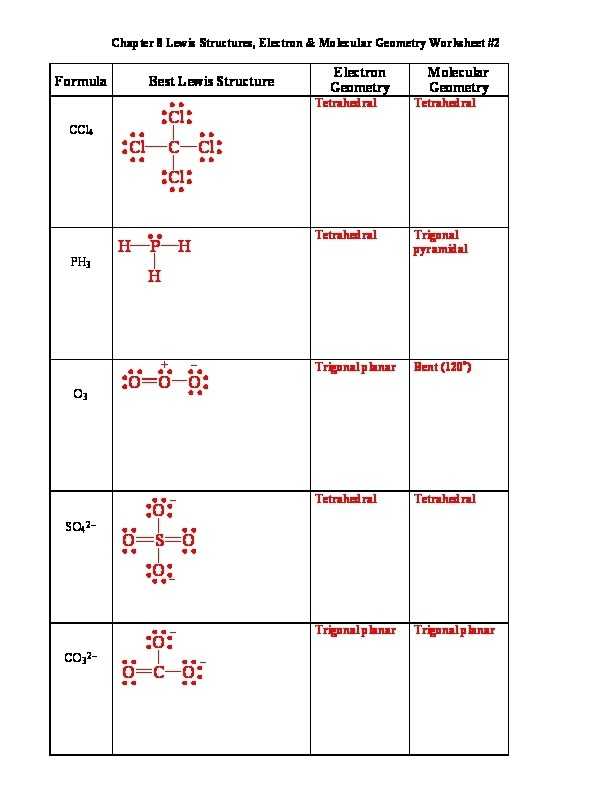
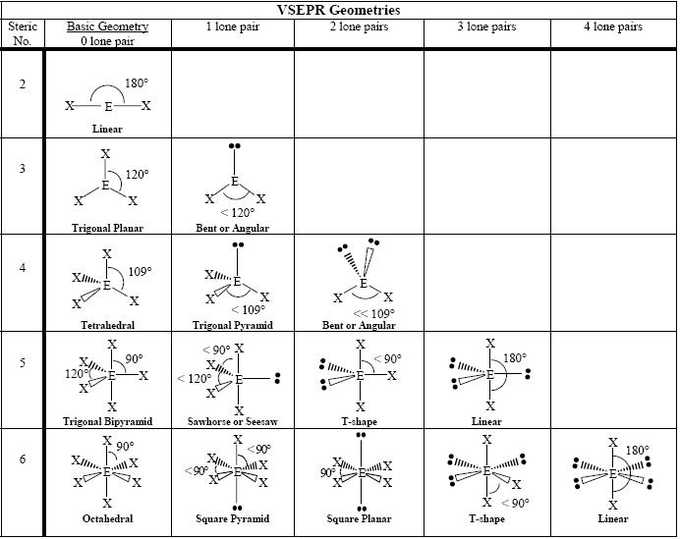
Molecular geometries consider the variety of atoms and the number of lone pair electrons. On the other hand, both geometries are related. They’re also known as the electron geometries. You are most likely already acquainted with these structures if you’re studying chemistry for some time. Don’t forget, formal charges can occasionally help you select which is the greatest Lewis Dot Structure whenever there is more than one possible structure.
The shape is known as a see-saw. It is called the octahedral. This shape is known as bent or angular. It is imperative to get accustomed to these common shapes so that you can establish the correct one.
The reactivity is affected in a lot of ways. Molecules will form certain shapes. These molecules can subsequently be employed to find out the geometry of the compounds. If you get a complicated molecule, break it down into smaller sections and appear at every atom individually. Once we include nonbonding electrons, that’s no longer correct. Nonbonding electrons have to be close to only 1 nucleus, and there’s a considerable quantity of space in which nonbonding electrons can reside and still be next to the nucleus of the atom. Quite simply, resonance doesn’t influence the shapes of molecules.
The issue of determining an accurate molecular geometry is quite complicated. Again the most typical mistakes are students getting confused about taking a look at the central atom to ascertain the range of bonded and lone pairs and students getting the incorrect Lewis Structure and thus the incorrect form. The appropriate answers are entered for you. Ensure you understand the reason why they are correct. So here is a direct cash flow program. Here you are at our site.

To use the VSEPR principle, one should compute a number called the steric number. To determine the form of the molecule, the variety of shared and lone pairs of electrons must be set. There are a lot of strategies to do a cash flow program.
At any moment, somebody will have several things as soon as it comes to short term and long term he wants to achieve, both. The aim is to challenge the worker to present his very best performance. The fundamental principle of the VSEPR theory is that molecular geometry can be predicted depending on the notion that electron pairs in molecules tend to repel one another and accomplish a maximum separation from one another. These are only a few of the basic postulates of the VSEPR theory that, alongside the lewis dot structure of a molecule, let us figure out the shape it’ll have. You can then use the Valence-shell electron-pair repulsion theory to ascertain how these unique tiny structures will connect to form a more complicated molecule.