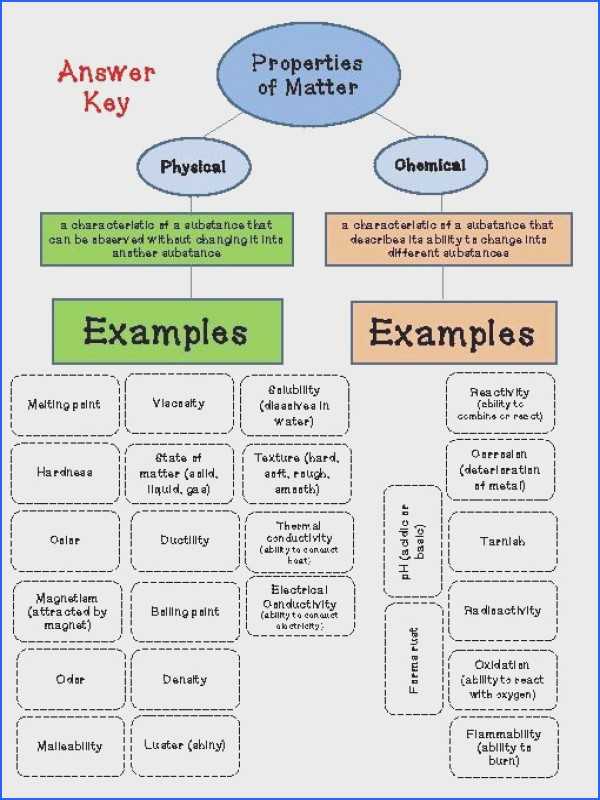
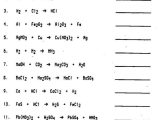Physical and Chemical Properties Worksheets is useful in helping students understand chemical properties of different compounds. The physical worksheet is used to give a chemical reaction step-by-step, including all the steps that a student needs to take in order to understand the chemical reaction. The chemical worksheet includes all the properties of the compound.
The Physical Properties Worksheet includes the amount of carbon dioxide produced, the amount of carbon monoxide produced, the amount of water required to make the compound, and the properties of the compound as determined by the student. The amount of carbon dioxide in a compound determines the gas law formula that will determine the amount of carbon dioxide in a mixture.
Carbon dioxide can be created from carbon, hydrogen, and oxygen. Each of these compounds has different amounts of carbon dioxide. The amount of carbon dioxide will affect the chemical reactions used to make the compound.
The amount of carbon dioxide will make it more difficult to add water to the mixture and will make it easier to lose some of the carbon dioxide through water vapor. When the amount of carbon dioxide is measured, the difference between the amounts will determine the reaction steps needed to make the compound. A simple example of a very simple compound is potassium nitrate.
The formula for potassium nitrate is CsNO3 + H2O = CO2. Therefore, it will take at least two steps to turn the carbon dioxide to carbon monoxide, or to remove the carbon dioxide from the compound.
The chemical properties worksheet allows students to determine the reaction with carbon dioxide in the step. Because the carbon dioxide can only be removed through water, it is very important to determine which combination of carbon dioxide and water is needed to complete the step. Students can also determine how much of the compound use to make up for the lack of carbon dioxide.

Students can include the carbon dioxide in their compound, or they can replace the carbon dioxide with hydrogen. When hydrogen is used, the student should take the ratio of carbon dioxide used into account when determining the amount of hydrogen used.
Complex chemical reactions can cause an increase in the amount of carbon dioxide, which can make it difficult to determine the amount of hydrogen needed to produce the compound. The physical and chemical properties worksheet will help students to determine the correct amount of carbon dioxide and hydrogen and to find out the most efficient ways to combine them.