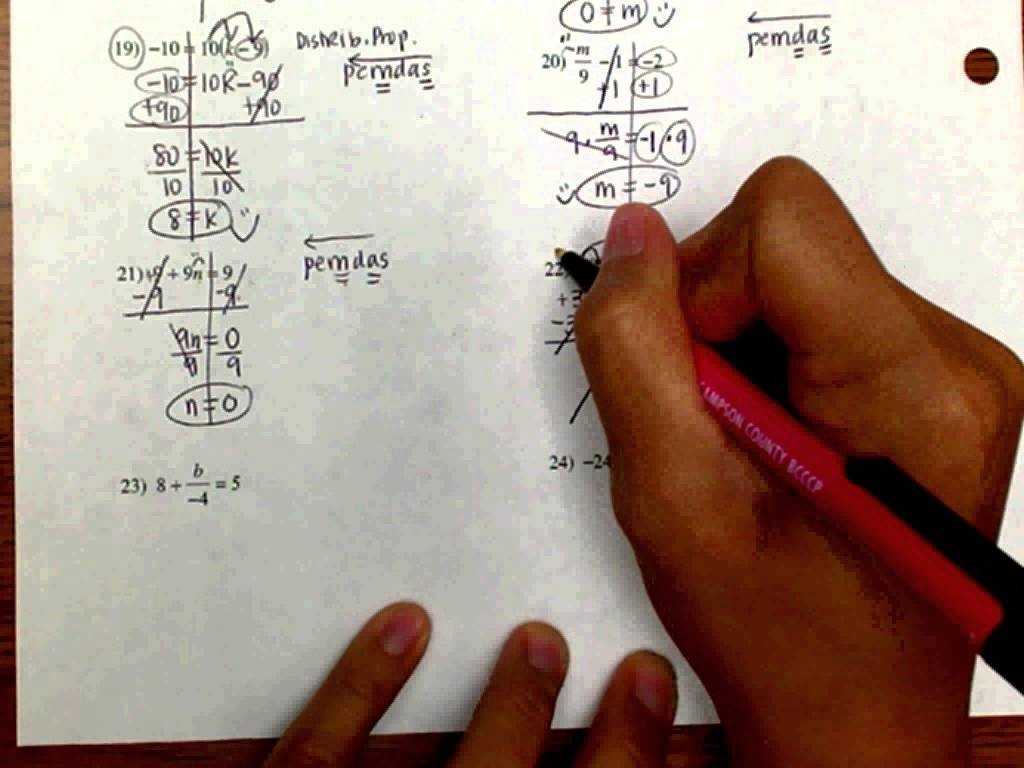Coulomb’s Law is an equation used to express the chemical bonding of molecules. It helps determine the amount of energy required to break bonds in the molecule. This form of energy is needed for the molecule to separate from one another.
There are several different ways to calculate this quantity. The common way is to multiply the molecular weight by the absolute number density. It is very easy to calculate the number density because the formula is quite simple. You just divide the mass of the molecule by the number of molecules that are to be split.
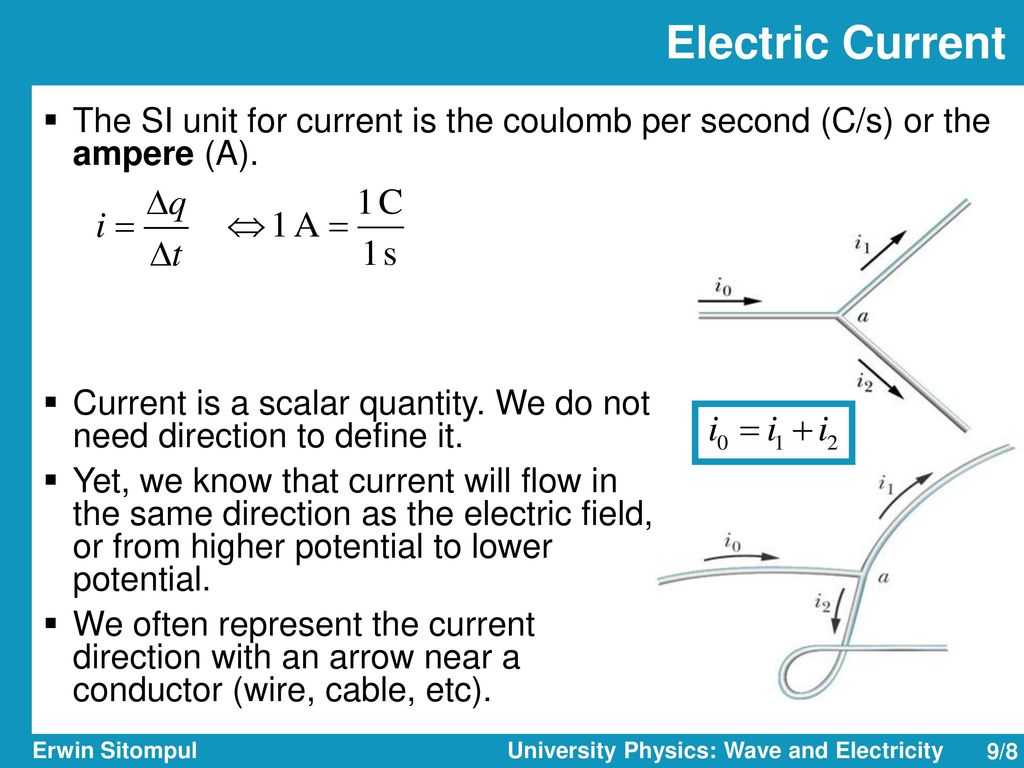
The base unit is the gram. The Greek system is used to calculate the weights. The most popular model for the Greek system is the foot. The compound weight is the number of grams that you need to split to equal one molecule. The answer to the question “What is the molecular weight of water?”
The moles of energy needed to break the bond is used for the rate of metabolism and reactions in cells. It is important to understand how the function of the molecule relates to these quantities. The enzyme that catalyzes the reaction is called a cyclic anhydrase. This part of the cell has two branches. The first branch makes oxygen and allows it to be produced by the process of photosynthesis.
The second branch has a ring on it that has three parts that look like a stamen and a bulb. This part of the enzyme has to have the right shape toorder to move in a specific direction so that the right process occurs. The weight of the water molecule would be less if the organelle was wider, but it would be more if it was smaller.
The second part of the enzyme is the electrochemical cell, which is responsible for the use of energy from the electron. The electrical potential energy depends on the binding energy of the oxygen atom. The reactions do not occur very quickly because the bonds between the molecules are not in very good shape.
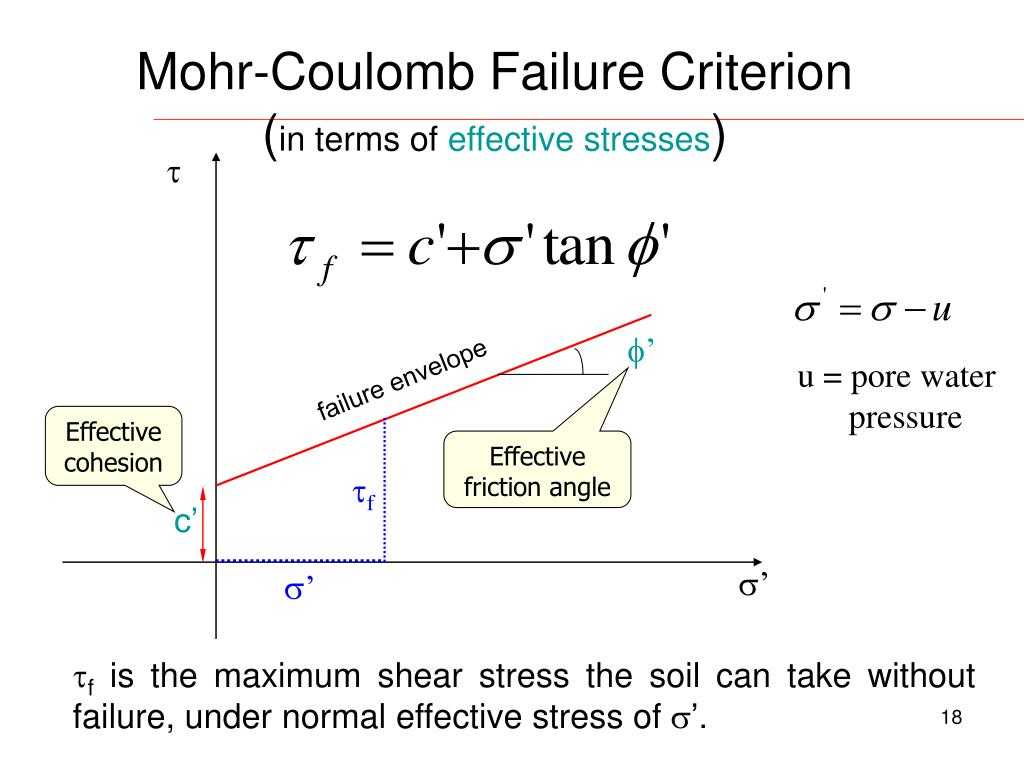
The moles of energy needed to break the bond are used for cellular metabolism and reactions. This makes it very important to learn how the energy needed for breaking the bond. It is important to know the electrochemical properties of the enzymes. When the moles of energy are needed to break the bond, the enzyme cannot absorb or release the electrons. The molecules will not be moved to the same place.
This is called a reversible reaction and will give you an efficient use of the energy released. This process allows the enzyme to break the bond, and the separation of the molecules will occur more slowly. The cells will not be as saturated with energy. If you take these factors into consideration in your class, you will find that you understand Coulomb’s Law better.