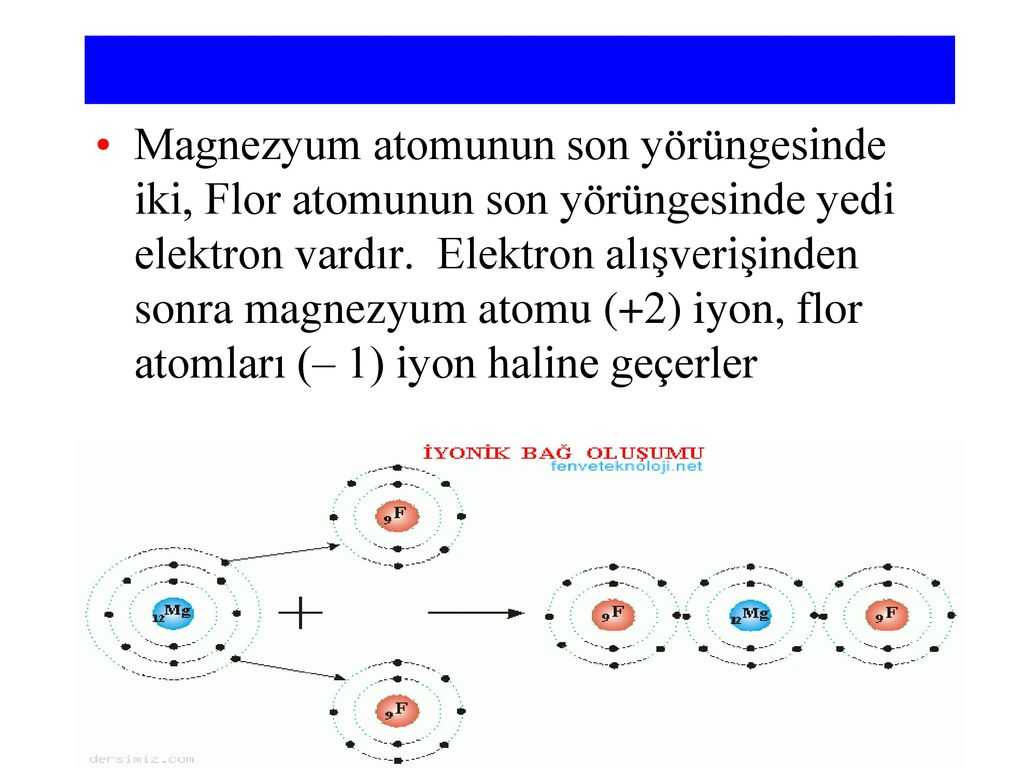
In a worksheet, atoms are arranged in rows and columns. The atoms are usually labeled A, B, C, D, etc. When they interact with the paper, an electrical current is generated.
It is easy to recognize that an atom is an atom because the direction of its electric field depends on the way it is oriented when it comes into contact with the surface. An atom can be represented as two parallel straight lines joined by a point. The two electrons in each line can also be of different kinds. Depending on how they are oriented, these electrons have different electric fields. Therefore, they create an alternating electric field.

By manipulating these atoms, you can change the orientation of the electrons. One way to do this is by adjusting the direction of their spin. In addition, when you rotate the atoms you get a different spin. You will find the opposite spins because when an atom is exposed to light, its electrons will change the orientation of their spin.
Isotopes are the same atoms that are present in atoms. Therefore, the molecules which are formed from these atoms are also the same. If the structure and composition of one molecule are similar to another, then these atoms will bind with the molecules.
Isotopes can be taken from your body. You will then need to split these atoms. You can choose from the various kinds of isotopes. It is important to know that each isotope has its own properties. It will be important for you to know the exact differences of each isotope.
There are isotopes of the elements Barium, Cesium, and Strontium. They will have their own characteristics and a unique ability to induce or reverse an electrical current. The structure of the atoms will also determine the rate of emission time.

Once you take one of these atoms, you will need to put it in a container. You can either split them yourself or have them sent to you. The container will need to be a container that has a lower atomic weight. The composition of the container will depend on the isotope you are going to use. The simplest composition for the mixture is a water-barium-sodium combination.
If you want to split the barium-sodium mixture, use a piece of silicon sheet. The composition can be easily determined by comparing the two compositions. Then you will need to allow for a few minutes for the mixture to cool before combining it into another container. Finally, you need to mix in one or two drops of iodine.